No, AI didn’t cure a dog’s cancer. But it helped.
A sick dog and the future of medicine
A Man and His Dog
Paul Conyngham was heartbroken. His dog Rosie had cancer. Thousands of dollars on chemotherapy and surgery hadn’t helped, and she only had months to live.
As a tech entrepreneur with decades of machine learning experience, Conyngham naturally turned to AI for help. He used ChatGPT as a research assistant, AlphaFold to analyze Rosie’s tissue, and Grok to suggest drug options. He partnered with three Australian research labs whose scientists did the hard work of sequencing Rosie’s genome and building a vaccine using the existing mRNA platform.
In December, Rosie finally got the shot along with a separate immunotherapy drug. Within weeks, her largest tumor had shrunk by 75% and she was back to chasing rabbits and jumping over fences.
The media pounced. Everyone loves dogs! Headlines like Newsweek’s “Owner With No Medical Background Invents Cure for Dog’s Terminal Cancer” celebrated the breakthrough.
Those headlines go too far in proclaiming a medical miracle. Rosie’s cancer is still terminal, and the AI tools Paul MacGyvered together only started a process that required professional researchers and expensive equipment.
From the garage to the lab
Clickbait aside, AI is making a real impact on drug development.
AlphaFold, the tool Conyngham used to analyze Rosie’s tissue, predicts the shape of proteins in 3D, identifying weaknesses drugs can exploit. AI then helps design molecules to target those vulnerabilities. Robotic labs are automating prototyping and AI toxicology may reduce the need for animal testing. Together, they are speeding up a design process that traditionally takes 5-7 years.
Personalized cancer vaccines are also being tested in humans. Merck and Moderna have spent the past five years developing a cancer therapy tailored to a person’s DNA. The drug is in Phase III trials, with early studies showing a 49% reduction in the risk of recurrence or death.
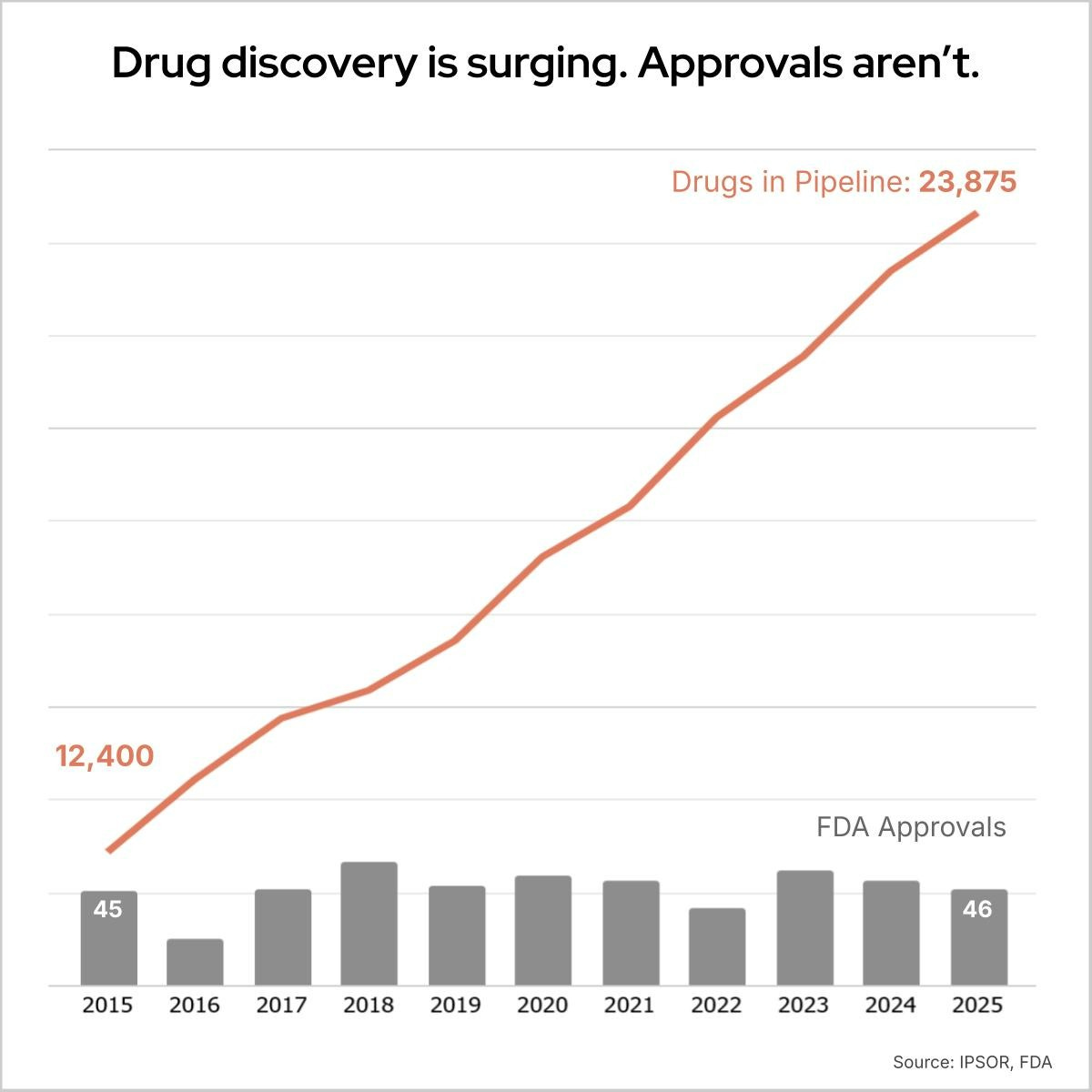
The global drug pipeline has more than doubled over the past decade. Now AI is adding fuel to the fire, with 30% of new drugs in 2025 discovered using AI.
But an overflowing pipeline doesn’t mean drugs reach patients faster. The biggest bottleneck is getting it approved. To understand why, we need to go back to 1960.
An unlikely hero
Dr. Francis Kelsey was born in Canada, studied at the University of Chicago and got her first job partly because the professor assumed “Frances” was a man.
In 1960 she was one of only seven FDA doctors reviewing drugs for safety. She was assigned to review an application for the morning sickness drug Thalidomide. The company expected fast approval, since the drug was already in use in Canada, Germany and 20 other countries.
But Kelsey was skeptical. The company didn’t have proof of safety, only patient testimonials and cherry-picked data. Instead of answering her requests, company representatives harassed her. They visited her office over 50 times, spread rumors that she was lazy and “pigheaded”, and even threatened to sue her.
Dr. Kelsey held the line. Overseas, patients miscarried or delivered babies with shortened or missing limbs. The drug was pulled from the market, but not before 10,000 babies were affected and thousands died.
In the U.S., it was only 17.
JFK awarded her the president’s medal for service, and the public outcry led to sweeping reform of US drug regulations, requiring clinical trials to prove both safety and efficacy.
The price of caution
In 2025, 46 new drugs were approved by the FDA. That’s the same number as in 2017.
The trials inspired by Dr. Kelsey are the gold standard for safety, but they’re slow. They require extensive patient and doctor recruitment, and typically take 7 or 8 years to reach FDA approval. Combined with discovery and design, most new drugs take 15 years to reach the market.
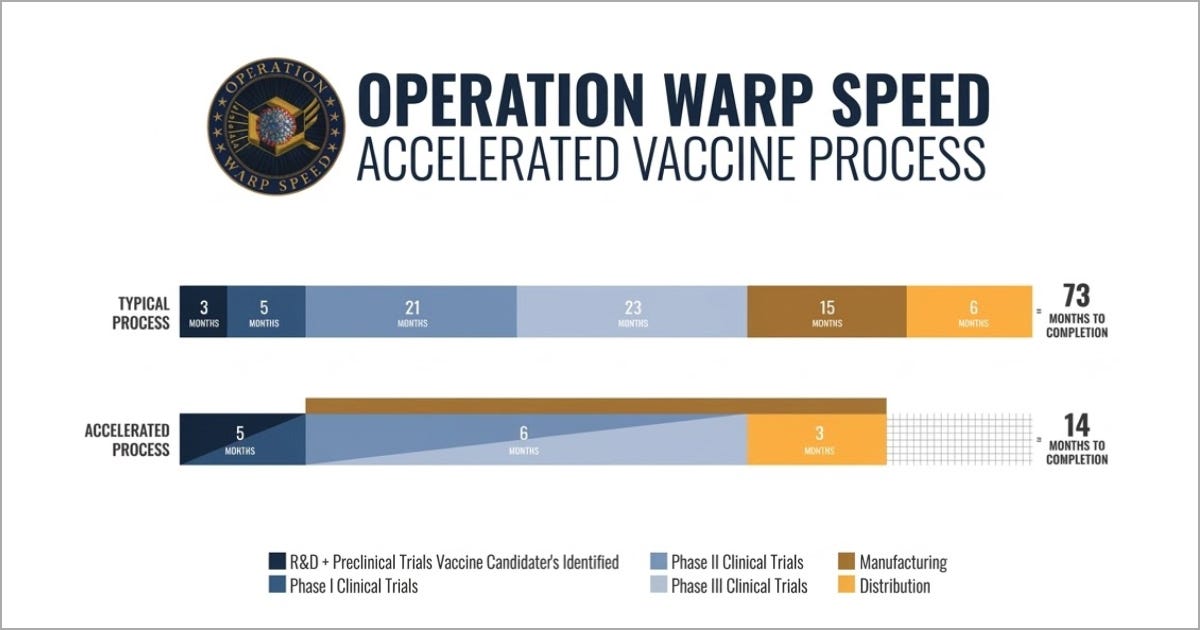
Covid forced America to rethink this process. Operation Warp Speed combined steps, expedited approvals and began manufacturing while testing was still underway. The first Americans were vaccinated only 11 months after the virus was identified.
Cancer isn’t Covid. You can measure vaccine efficacy in weeks, while cancer takes years. But Warp Speed showed that urgency and rigor can coexist, and the FDA launched a pilot program in 2025 called START to fast-track approval decisions for rare diseases.
The headlines that aren’t written
Dr. Kelsey is a hero. And while the drug testing process she inspired saves lives, it also costs them.
Statins like Lipitor and Crestor save an estimated 50,000 lives each year. Back-of-the-envelope math says that 4,000 people died every month they weren’t approved.
The questions are agonizing: Should we accelerate approvals knowing some drugs will save lives and others will harm or kill people? Or should we go slowly to maximize safety, knowing patients will suffer and die while they wait?
AI will make these questions more urgent. Moderna’s personalized cancer vaccine showed positive results in 2022, but the FDA declined to expedite its approval. It will reach the market in 2027 at the earliest.
Lives lost to harmful drugs make headlines. Ones lost to approval delays don’t.
Dad Joke:
Why did the sick dog go to the AI lab? She wanted to “retriever” test results. 😭